- Get directions
- Leave a review
- Claim listing
- Bookmark
- Share
- Report
- prev
- next
- Monday, May 2, 2022 @ 10:00 am
In 2021 the global biotech industry was again performing at new heights, considering the worldwide launch of various new vaccines as protection against COVID-19, as well as therapeutics to address COVID-19. Companies like the German BioNTech (together with Pfizer) and the US company Moderna (with strong support from the Swiss CMO Lonza) were able to develop and launch new mRNA-based vaccines in record time.

Frederik Schmachtenberg
EY | Partner, Global Health Sciences & Wellness Lead for Financial Accounting Advisory Services
Global biotech landscape
Biotech IPOs continued to be “en vogue” although the number of SPAC transactions started to slow down somewhat in 2021. The IPO class of 2021 recorded a total of 143 IPOs (2020: 73), which generated more than USD 19.3 billion in funding (2020: USD 11.2 billion). The significant increase in IPOs can mainly be attributed to the high count of US SPAC transactions. 99 US IPOs (2020: 63) were able to collect a total of USD 15.7 billion (2020: USD 10.5 billion). In Europe, biotechs successfully completed 44 IPOs (2020: 10), generating USD 3.6 billion (2020: USD 0.7 billion).
Swiss biotech landscape
In 2021, the Swiss Biotech industry saw a record-high revenue figure of CHF 6.7 billion (2020: CHF 4.9 billion). Such significant uptick was mainly driven by an increase in product sales, favorable one-time events from collaboration and licensing deals, as well as a general positive advancement of the product pipeline and, as a result of that, regulatory approvals continuing at very high levels (also refer to the product development section below).
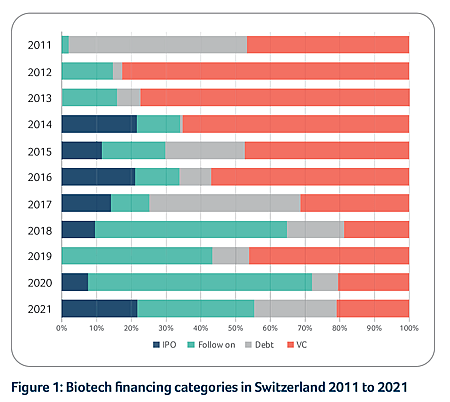
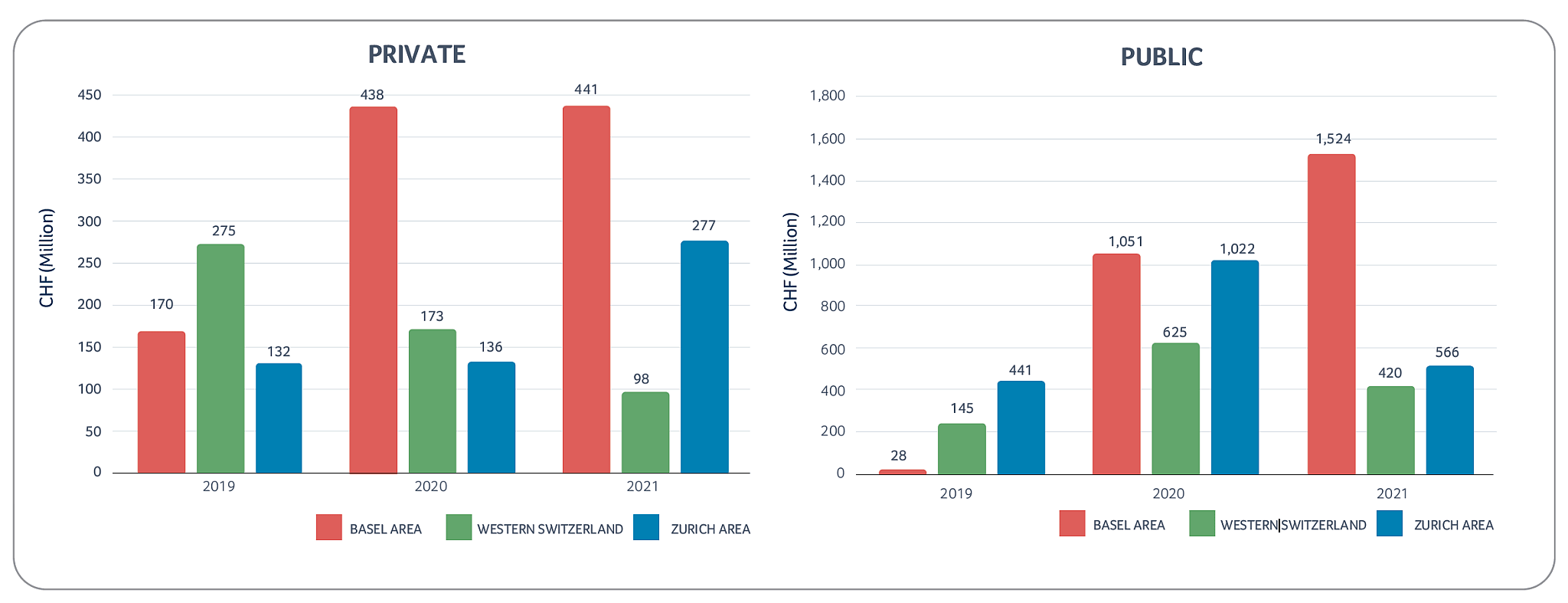
Swiss biotech financing
The Swiss biotech industry was able to raise more than CHF 3.3 billion in 2021, with roughly CHF 2.5 billion collected by public companies and the remaining CHF 0.8 billion collected by private companies. Anaveon and Numab Therapeutics recorded the largest private company financing transactions in 2021, with CHF 110 million and CHF 100 million raised, respectively. Furthermore, several biotechs benefited from cantonal COVID-19 loans as well as the COVID-19 innovation program financed by the Swiss government.
A number of successful European IPOs were also announced in 2021, five of which either involved Swiss companies or used SIX Swiss Exchange as a platform. NLS Therapeutics, VectivBio, and Sophia Genetics all had their public market debut on Nasdaq during the 2021 spring/summer timeframe. The latter two were even able to execute successfully their “green shoe”. Schlieren-based Molecular Partners did a secondary listing on Nasdaq in the 3rd quarter of 2021. SIX Swiss Exchange took note of the successful IPO of PolyPeptide Group in the first half of the year and Idorsia also benefited from the favorable financial climate and placed a CHF 600 million convertible bond on SIX Swiss Exchange at the end of July 2021. An additional transaction noteworthy on SIX Swiss Exchange was the reverse merger transaction between Polyphor and EnBiotix (a private US biotech), which led to the creation of Spexis as a new company, assuming Polyphor’s spot on the trading floor in Zurich.
Last but not least, in late 2021 the recently founded company MoonLake Immunotherapeutics also started its preparation for an upcoming SPAC transaction on Nasdaq.
In spring 2021, Pureos Bioventures announced the final closure of its first fund BB Pureos Bioventures LP with committed capital of USD 205 million. Pureos will exclusively invest in innovative private drug development companies with an emphasis on novel biological drugs and emerging modalities, such as nucleic acid, and cell and gene therapies.
M&A and collaborations
Swiss companies were involved in several significant M&A transactions:
- Mestex AG was acquired by Grünenthal Pharma
- Relief Therapeutics acquired Applied Pharma Research
- Novartis took over Cellerys
- Atlas Antibodies purchased the biotech service provider evitria
- Novaremed bought Metys Pharmaceuticals
- Inositec was acquired by Vifor Pharma, which itself was later acquired by CSL Behring
- Alloy Therapeutics acquired deepCDR Biologics
- Merck acquired Chord Therapeutics to expand its neuroinflammatory pipeline
Also, entering into license and collaboration agreements continued to be “en vogue” in 2021. A selection of such transactions is mentioned below:
- Lonza/Moderna entered a new agreement to double drug substance capacity production for Covid-19 in Visp
- Deep Breath Intelligence collaborated with Bayer
- Genedata/Sanofi extended their collaboration for pharmaceutical development & manufacturing science
- Organon/ObsEva entered into a global license agreement to develop and commercialize ebopriprant
- Molecular Partners/Novartis signed a collaboration to develop DARPin-conjugated radioligand therapeutic candidates for oncology
Product developments
In 2021, the industry saw a similar number of regulatory approvals compared to prior years. More specifically, EMA approved 91 new drugs in 2021 (2020: 97 drugs) and the FDA approved 50 new drugs (2020: 57 drugs), not counting the emergency use approvals for therapeutics in the fight against COVID-19, one of which originates from Humabs in Bellinzona.
Furthermore, among the new FDA approvals there were three new drugs which the Swiss biotech sector can proudly claim as “originated in Switzerland”, namely ADC Therapeutics’ Zynlonta, Adumhelm from Biogen/Neurimmune, as well as Ponvory from J&J/Actelion/Idorsia.
The count of drug approvals by Swissmedic exceeded the prior year with a total count of 45 new drugs (2020: 42). However, and not unusually for the sector, some setbacks in clinical developments also had to be noted in 2021, for example with Polyphor’s late-stage breast cancer study failure, which paved the way for the above-mentioned reverse merger transaction.