- Get directions
- Leave a review
- Claim listing
- Bookmark
- Share
- Report
- prev
- next
- Tuesday, April 25, 2023 @ 7:00 am
Global advances in biotechnology and personalized medicine pose a challenge to regulatory authorities. Patients expect to benefit as quickly as possible from innovative and effective therapeutic products and treatments, but these must be safe and of high quality.

Jörg Schläpfer
Swissmedic | Head of Management Services and International Affairs
Swissmedic strengthens the Swiss research hub by supporting innovation and accelerating the approval process for new products long before they are ready to be commercialized. Through collaborations with international partners, Swissmedic facilitates controlled and rapid access to promising products for drug developers and patients not only in Switzerland but around the world.
The role of Swissmedic
Swissmedic was established in 2002 and over the past two decades it has had a major impact on the implementation of Swiss innovation to protect human and animal health around the globe.
As the Swiss authority responsible for the licensing, authorization and monitoring of medicinal products, Swissmedic is involved in the entire life cycle of medicines. Efficient assessment of advanced therapy medicinal products (ATMPs) requires Swissmedic staff to constantly renew and expand their competencies.
The COVID-19 pandemic demonstrated the importance of intensive dialogue with research groups and applicants before and during the review procedure, and made a vital contribution to fast – but carefully considered – authorization decisions.
Swissmedic innovation office
While Swissmedic is learning from the experiences of innovation offices at other regulatory authorities, it also wants to shape its own solution – the version that is appropriate for Switzerland.
Created at the end of 2022, the innovation office is a new concept in Swissmedic’s portfolio and is designed to achieve faster access to innovative medicines. The idea is to take a step closer to the innovators so that small companies and startups can be supported from an early stage.
The innovation office promotes networking and regular dialogue with research organizations and industry in order to become closer to the drivers of innovation and to tackle regulatory challenges in a solution-focused manner. During an initial pilot phase, it will concentrate on small operators – on the startups, spin-offs, research groups and incubators that promote innovation.
Low-threshold advice services are designed to support those who have concerns about contacting the regulator. The sooner they know what is needed, the faster a successful outcome can be achieved. The earlier a mistake is discovered, the more easily it can be corrected, so that neither the medicinal product nor the startup is jeopardized.
Lessons from the COVID-19 pandemic are also being implemented. During the pandemic, active dialogue with stakeholders and access to scientific advice and support were crucial to allow COVID-19 vaccines to be authorized at unprecedented speed. Moreover, scientific advice for clinical trials was offered for the first time during the pandemic, and in many cases this enabled the required licenses to be granted within a few days, particularly in the case of science-initiated studies (investigator initiated trials).
New ways of connecting with target groups to provide access to expert advice, and new ways of sharing information are currently up for discussion. Universities and innovation hubs are approached proactively and “regulatory speed dating” may help startups overcome possible preconceptions about the “medicines police in Bern”. Initially, the innovation office will restrict itself to advice relating to ATMPs, since Swissmedic is able to act most flexibly in this area. After a pilot phase that is expected to last from 18 to 24 months to determine the appropriate scale and the most effective form of cooperation, Swissmedic would like to extend its accumulated experience to other therapeutic products.
By identifying and supporting highly promising medical innovations at an early stage and working with other research organizations, Swissmedic will contribute to the success of Switzerland as a leading global research hub and help those patients for whom adequate treatments do not yet exist.
Innovating Swissmedic’s own procedures
Swissmedic is increasingly transforming itself from a documentdriven to a data-driven organization. Core processes have now been digitized and our highly integrated IT systems allow timescales to be adhered to or shortened. Interactions will soon be processed via a new portal which allows applicants to submit their data in a structured manner, thereby optimizing the ability to plan ahead and simplify application planning. The reorganization of operations and modified processes also form the basis of a more agile approach to regulation so that decisions can be made more quickly but with no reduction in rigor.
New technologies are opening up other highly promising fields in relation to the use of artificial intelligence (AI) and machine learning to assist in processing large volumes of data provided as supporting evidence. The pharmaceutical industry already employs systems that sort and initially assess adverse drug reactions using AI.
It will probably not be possible in the foreseeable future to review applications exclusively with a system based on AI as the number of currently available marketing authorization applications and authorization decisions from the past do not provide a sufficient basis for “training” AI systems, and decadesold authorization dossiers cannot help an AI system to evaluate today’s applications. But intelligent technologies could be used in other areas to help refine, search or contextualize data, and automate standardizable and repetitive tasks. These functions are crucial in the areas of authorization, market surveillance and licensing for central processes, for example in the preparation of certain licenses and assisting in the processing of adverse drug reaction reports.
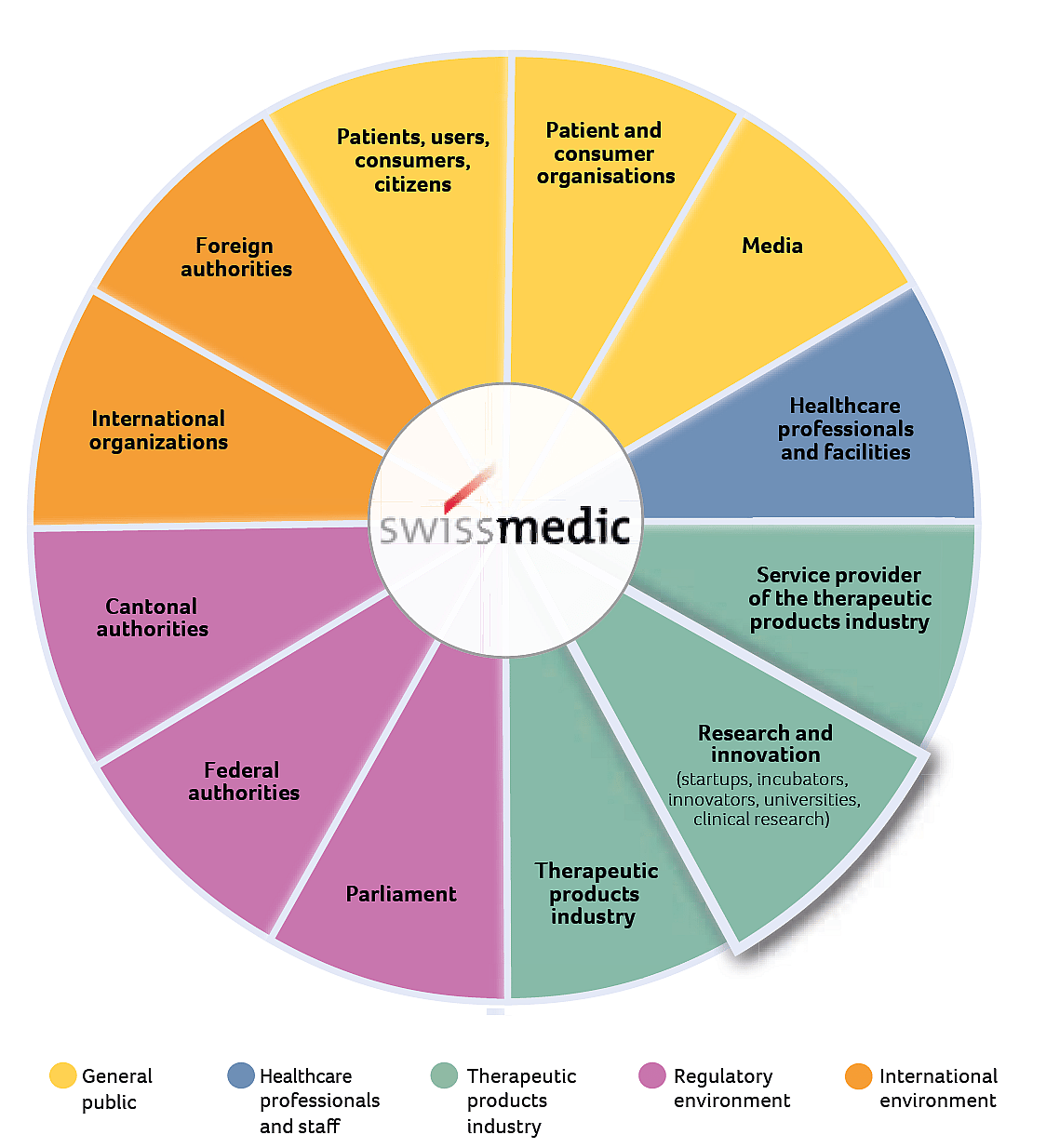
Swissmedic Stakeholder Map
Innovation as a focal point of Swissmedic’s stakeholder engagement
The key to successful innovative solutions
The therapeutic products sector thrives on innovation and Swissmedic must establish itself as an innovation enabler. With the compassionate use and the temporary authorization of human medicinal products it already has several options to support rapid access to innovative medicines.
Swissmedic also offers scientific advice or pre-submission meetings, not just for authorization applications but also, for example, for establishment licenses for new production facilities or for situations in which researchers have to decide the best study design for their new active substance at an early stage of drug development.
By interacting with research organizations, Swissmedic can keep abreast of the latest technology trends and understand envisaged solutions. Applicants, for their part, receive regulatory feedback and valuable know-how to help them advance their innovation or infrastructure projects.