- Get directions
- Leave a review
- Claim listing
- Bookmark
- Share
- Report
- prev
- next
- Tuesday, May 5, 2026 @ 11:00 am
Swiss-based scientists Christian Klein and Pablo Umaña have been working closely together for over 20 years and are distinguished by both the number and quality of their inventions in biotechnology. The following interview provides insights from the perspective of this biotech inventor dream team. Their shared area of interest is antibodies for immunotherapy, with a focus on bi- and multispecific antibodies.



Pablo Umaña
Pablo Umaña is a chemical engineer and molecular immunologist from Costa Rica. He obtained his PhD at Caltech and followed his professor to ETH Zurich in the mid-1990s. He is one of the co-founders of the company Glycart Biotechnology, acquired by Roche in 2005. Pablo continued to work for Roche Glycart until 2024 and is now co-founder and CEO of a biotech startup company in Zurich.
Christian Klein
Christian Klein is a biochemist from Germany. He started to work for Roche in Germany in 2002. The acquisition of Glycart was the starting point of his collaboration with Pablo. Christian moved to Switzerland in 2009 to join Roche Glycart, where he remained until 2024. After working with Curie.Bio as a CXO he is now co-founder and CSO of a biotech startup company in Zurich. He also acts as an external lecturer in biochemistry at LMU in Munich.
"A patent is a contract you make with society. You get the protection for a certain time, in exchange for teaching exactly how to carry out a specific procedure, which in turn allows the field to move faster."
— Pablo Umaña
"Some of our inventions may not have panned out in an approved drug, but they have advanced the field by inspiring others to find alternative or better solutions."
— Christian Klein
Introduction
Between 1994 and 2023, nearly one million patent families were filed worldwide in the field of biotechnology. Of these, around 11,000 patent families list inventors with residence in Switzerland.
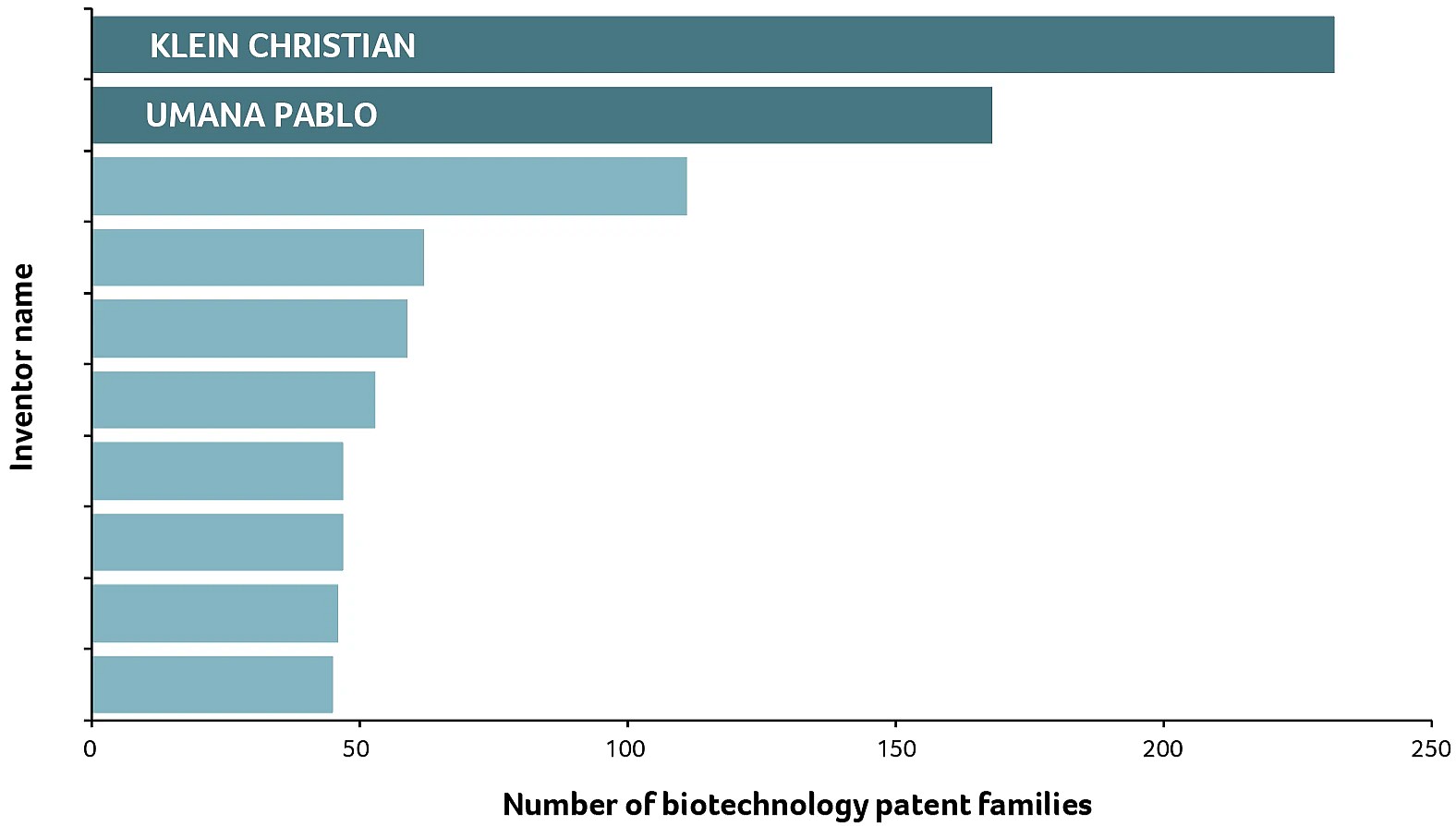
Among these patents co-invented in Switzerland, Christian Klein and Pablo Umaña stand out as the two inventors with the highest number of filings (Figure 1). Their names appear as inventors of almost 300 patent families.
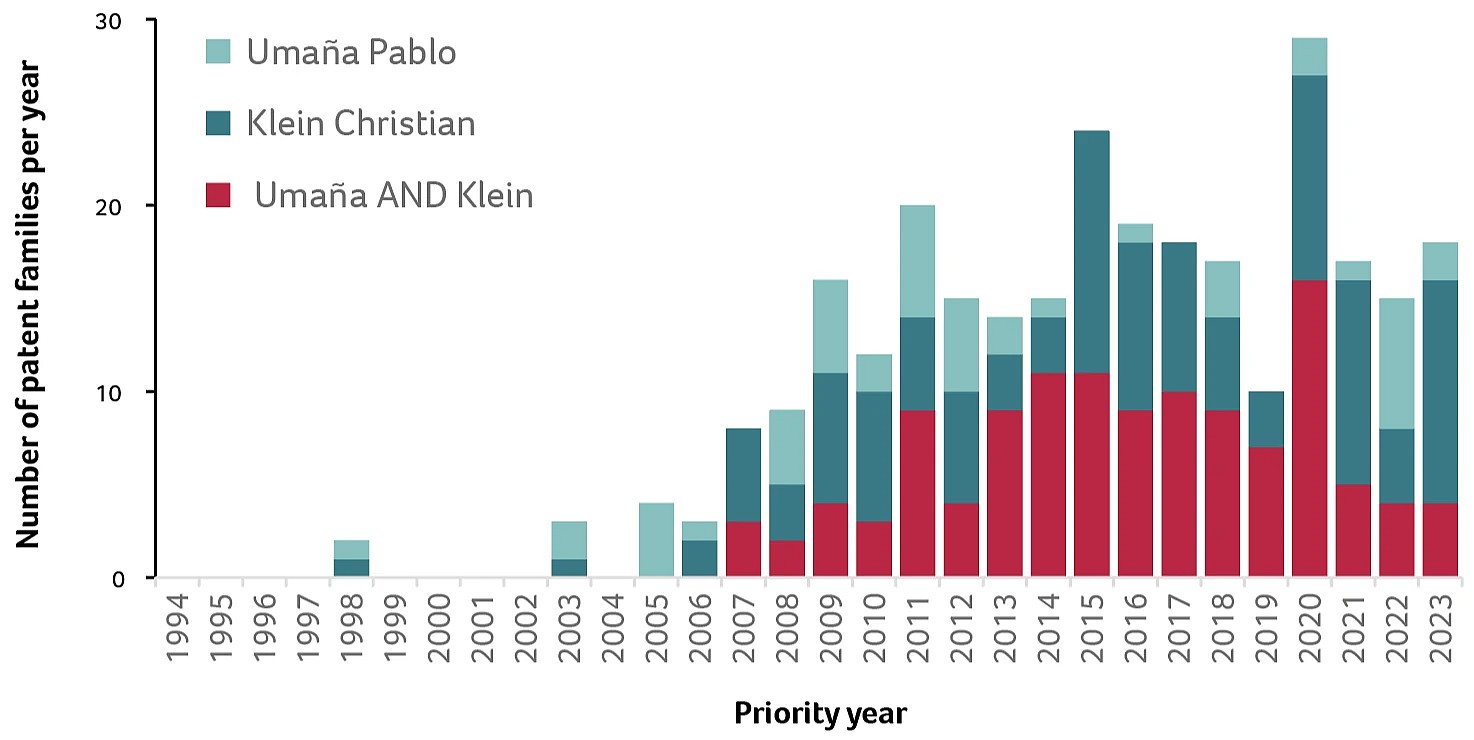
The patent data reveals the start of their collaboration, with their first joint priority filings in 2007, followed by multiple filings every year until 2023 (Figure 2).
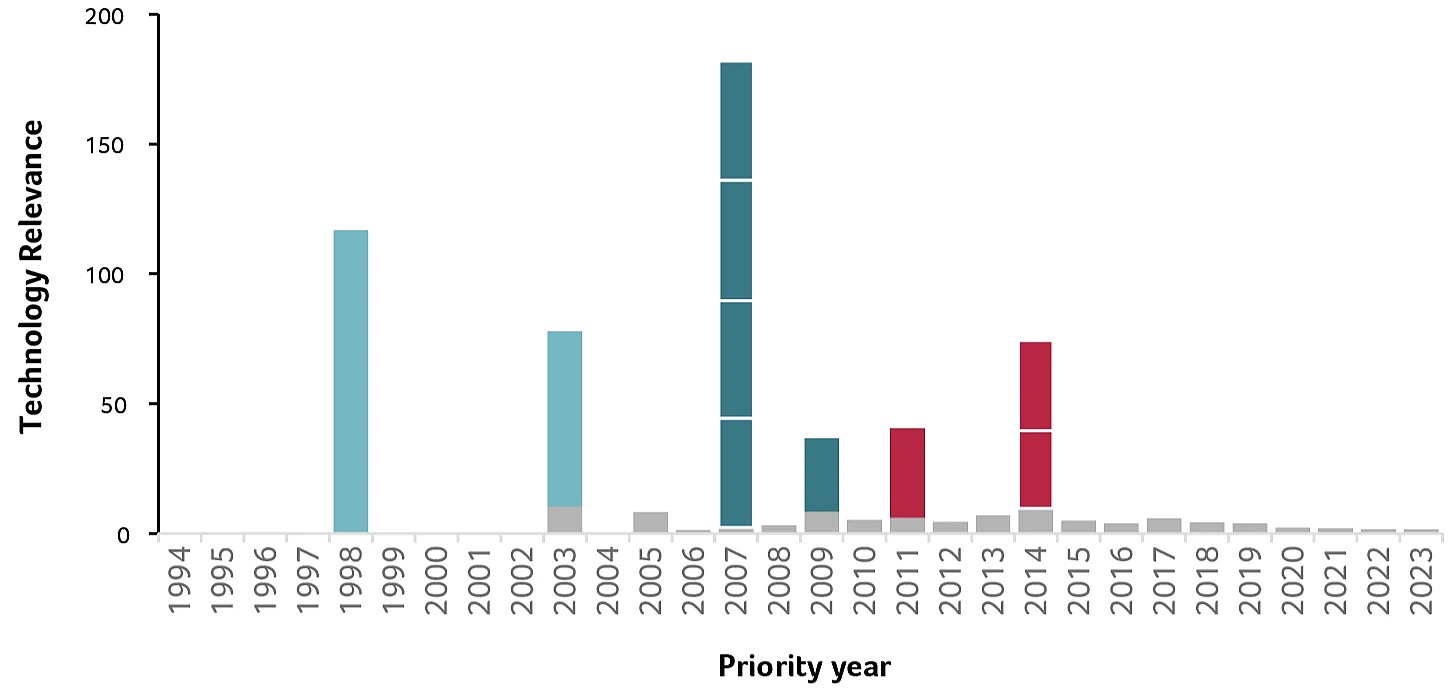
Christian Klein and Pablo Umaña also excel in terms of the quality of their inventions, measured by Technology Relevance™ (TR), an indicator based on citations. The average TR score of their 300 patent families is 6, compared to an average of 1.9 for all biotech patents from Swiss inventors, and 1 for all biotech patents worldwide (Figure 3).
They made their first high-impact inventions independently of each other: Pablo Umaña filed patents in 1998 and 2003; Christian Klein had filings in 2007 and 2009 while based in Germany. Three more high-impact families with TR scores over 25 followed in 2011 and 2014, with Pablo and Christian as co-inventors working for Roche-Glycart in Switzerland.
Interview
Looking at the patent statistics, the two of you are named as inventors of approximately 300 patent families, and you appear as co-inventors in over 120 of these families. How did you become such productive serial inventors?
PU: We are fortunate that we are passionate about our work. We are constantly thinking about relevant challenges, and try to come up with solutions, both inside and outside working hours.
CK: Challenging the status quo and thinking how can things be further improved is what drives us. We are always attracted to the next challenge, not doing a "me-too" kind of thing, but going a step further to develop first-in-class or best-in-class approaches by addressing key questions, even if they appear very difficult in the beginning. At the same time, we need to achieve the right balance by finding solutions that are realistic and can be developed as drugs.
Your track record is even more impressive when looking at quality indicators of your patents. We have identified 10 patent families of particularly high Technology Relevance™.
CK: These scores correspond to what we think are the most relevant patents we have contributed to, for example bispecific antibodies, or cis-targeted cytokines. These patent applications had an impact on how the field developed. You can see that nowadays there are numerous bispecific antibodies using related approaches in clinical development.
The big question in the context of inventions often is: how did the inventors come up with this? Inspiration out of the blue? Hard work towards a goal? Or surprising results that overturn the working hypothesis?
CK: In reality, it is a mixture of all three. There are some inventions that really came out of the blue, but there are many others that are building on what has been done before, finding very specific and new solutions.
PU: The unexpected result itself may be the invention. A surprising finding may make you question all your assumptions. You realize that there is value in attacking the problem in a different way.
When advancing in your career, you probably had increasingly managerial functions rather than being at the bench. How important is the team during the creative process that leads to inventions?
PU: We have always been surrounded by very good teams, which enabled us to collaborate with exceptionally productive and expert colleagues. When no longer working at the bench, both of us still tried to stay very connected to the science and to remain involved in the details of the projects.
CK: But we are not speaking about micro-management, more about the conceptual point of view.
PU: Absolutely. Discussing science is something we like and are good at, so we try to keep that as a component of what we do every day.
When thinking about challenges to solve, what is your starting point?
PU: Our focus is always patients in need of better treatments. Then we think about molecules which might work — first based on our understanding of the biology and then considering protein engineering approaches.
CK: For example, a bispecific antibody can provide a big advantage to the patient for certain diseases. At the same time, we have to think about how to achieve best efficacy with minimal side effects. We will not start developing a fancy technology and only later look for a potential application.
While highly specialized in your main topic of interest, you are also generalists, with the big picture in mind.
CK: That probably explains our productivity to some extent. You may have people who are pure antibody engineers with limited understanding of where their science can be best applied. And then you may have people focused on the biological or clinical aspects, but they lack the insights on how you can make a molecule. It is crucially important to combine both sides.
PU: Ultimately, you need a deep understanding of all aspects of the problem you are trying to address: disease biology, biochemistry, engineering, and drug development. Speed is important too: you need to assess very rapidly the feasibility of your ideas. It works very well with the two of us because we both have this background. Many of our inventions are the result of ideas that bounced between Christian and myself. Of course, this creative process of developing ideas also involves our teams.
Developing a drug is a very long process, starting from ideas to treating real patients. Can you give us an impression of how many ideas you have had to generate these 300 patent families, and what it represents in terms of patient treatment?
CK: It is true that many ideas will never even reach the patent filing level, because they are not feasible, not realistic. Our entire work has generated over 30 drug candidates which have reached the clinical development stage, and at least three of them are approved products for diverse diseases now. Hopefully, a few more will follow in the future.
How much do you think about the patentability of an idea during your development process?
PU: Of course, we have to think about patents from the beginning, because a new product requires significant investment, and to secure this investment you need to obtain a patent. Without a patent, the product will never be developed. That is the reality in the pharma industry.
CK: But on the other hand, the patents will expire after 20 years and everyone is then free to produce biosimilars or generics. That is quite unique. A musician, for example, will keep the rights on his compositions for his entire life and beyond.
You both worked in academia early in your careers and then switched to industry. How do those two worlds differ?
CK: In industry, the primary goal is to develop new products, and the patent is the must have. In academia, the publication is the must have, and the patent is often seen as nice to have. But that means that universities probably miss certain opportunities, because nobody recognizes the value of their inventions.
PU: In academia, you have the same intention to advance the field as we have in mind in industry, yet the focus is completely different.
CK: Let's take the example of a very fancy discovery like CRISPR. You are aware of your academic competitors, so you want to publish before them. In industry, we would take the opportunity of the 18-month period between filing and publication of the patent to maximize the impact of the patent. In academia, scientists do not have time to wait for 18 months.
Based on your experience, what suggestions and advice can you give to young researchers at the beginning of their careers? In general, and in particular with regard to inventorship and patenting?
PU: If you want to be an inventor, you must be passionate about the field you are working in and you need a strong problem-solving mindset. You should also be oriented towards practical applications. You need to stay up to date, to be continuously learning, for example by studying the literature, including patents. Also, you have to be open to collaboration, because team work and interdisciplinary interactions are key.
CK: Sitting alone and doing your things on your own will limit what you can achieve. In the end, you require a team of motivated people with complementary expertise to work jointly on novel solutions.
PU: At least initially, for the conception, you need to have a small group or at least a partner to bounce ideas back and forth, to challenge the ideas. This kind of discussion is an important part of the creative process. It really helps you to improve the initial ideas, rather than just thinking in isolation in a vacuum.
IPI would like to thank Christian Klein and Pablo Umaña for sharing their insights with readers of the Swiss Biotech Report.