- Get directions
- Leave a review
- Claim listing
- Bookmark
- Share
- Report
- prev
- next
- Tuesday, May 5, 2026 @ 11:00 am
Switzerland's strength as a global biotech hub is often described in terms of innovation, productivity, and quality. At the heart of all three lies a decisive factor: talent. From early scientific discovery to large-scale manufacturing and global commercial leadership, Switzerland offers a talent ecosystem that supports the entire biotech value chain.

Sirpa Tsimal
Switzerland Global Enterprise | Director Investment Promotion
In this article, Sirpa Tsimal talks to Carl Deutsch of NBE Therapeutic to gain insights into how the Swiss research and talent ecosystem supports scientific discovery, translational research, and the development of next-generation therapies in oncology. She considers why the availability of specialized talent is also a vital ingredient for the manufacture of biologics and advanced therapies for global markets. In the final section Sirpa speaks to Alexey Kutahov of Mirum Pharmaceuticals, who explains how Switzerland supports global launches, leadership development, and long-term talent retention.
Discovery and R&D — Interview with Carl Deutsch, Chief Scientific Officer, NBE Therapeutics
At the discovery and R&D end of the biotech value chain, Switzerland offers a distributed talent environment spanning several regions. The Basel area plays a central role, supported by strong academic and applied research hubs in Zurich, Lausanne, and other regions, enabling close interaction between industry, research institutions, and clinical partners.

Could you briefly outline NBE Therapeutics' core scientific and R&D focus areas?
NBE Therapeutics focuses on advancing next‑generation antibody-drug conjugates (ADCs) for oncology. ADCs are a special type of antibodies that deliver a chemotherapy drug (also called a payload) directly to tumor cells. Since becoming part of Boehringer Ingelheim in 2020, we've combined our proprietary payload and antibody-conjugation technologies with the expanded scientific capabilities available through the pharmaceutical company. This collaborative setup allows us to design ADCs with precision, developing targeted therapies that aim to deliver potent treatments directly to cancer cells while sparing healthy tissue.
Our team contributes to a growing ADC pipeline designed to bring first‑ and best‑in‑class solutions to patients with difficult‑to‑treat cancers. The integration of NBE's deep expertise with Boehringer Ingelheim's translational and clinical strengths creates a powerful engine for innovation in precision oncology.
What were the key reasons for establishing NBE Therapeutics' R&D operations in Switzerland, and how did access to scientific and technical talent influence that decision?
NBE Therapeutics chose to build its R&D operations in Basel because the region offers an unusually rich environment for biotech innovation. Basel has one of Europe's most concentrated life sciences ecosystems, where global pharma companies, growing biotechs, and ambitious startups sit practically side by side. This creates a culture of collaboration, fast learning, and easy access to specialized infrastructure that young companies rarely find elsewhere.
Just as important is the talent pool: Basel draws scientists and technical experts from Switzerland's top universities, from across the border in France and Germany, and increasingly from further afield. The city's international character, strong academic networks, and cross‑border mobility make it an ideal place to attract and retain world‑class researchers. Together, these factors make Basel a uniquely compelling home for NBE Therapeutics' R&D engine. Furthermore, this environment not only supported NBE's growth but continues to strengthen our role within Boehringer Ingelheim's European R&D footprint.
From your perspective, what specific strengths does Switzerland's life sciences ecosystem offer when it comes to attracting, developing, and retaining top-tier R&D talent, such as scientists, translational researchers, and technical specialists?
Switzerland combines scientific excellence and expertise with an exceptional quality of life. The country is home to world‑class universities and research institutes, supported by strong biotech–pharma clusters in Basel, Zurich, and Lausanne that provide scientists with diverse career opportunities. What sets the country apart is its seamless infrastructure: short distances between academic hubs, efficient public services, and well‑established networks that connect researchers across institutions and industries.
This combination creates a melting pot for new ideas, fosters collaboration, and promotes innovation.
On top of the infrastructure, a stable and reliable regulatory framework, including strong IP protection and competitive salaries make Switzerland an attractive place for scientists. It is important to note that all of this is made possible by highly efficient and supportive public and governmental services, which make Basel, and Switzerland as a whole, a truly attractive environment for scientific careers.
What is NBE Therapeutics' broader European and international growth strategy?
NBE Therapeutics has played a strategic role in shaping Boehringer's ambitions in next‑generation ADCs. Our scientific and clinical expansion, including the establishment of a new ADC R&D center in Basel, is fully aligned with Boehringer Ingelheim's global oncology strategy and supported by its broad research infrastructure, partnerships and development expertise.
Together, we are growing an integrated ADC capability, from discovery to clinical development. This includes partnering with top external innovators (e.g., Synaffix, AimedBio) and building the specialized facilities and teams needed to accelerate breakthroughs. Several ADC programs are already advancing through the pipeline, reflecting the shared commitment to bringing transformative therapies to patients worldwide.
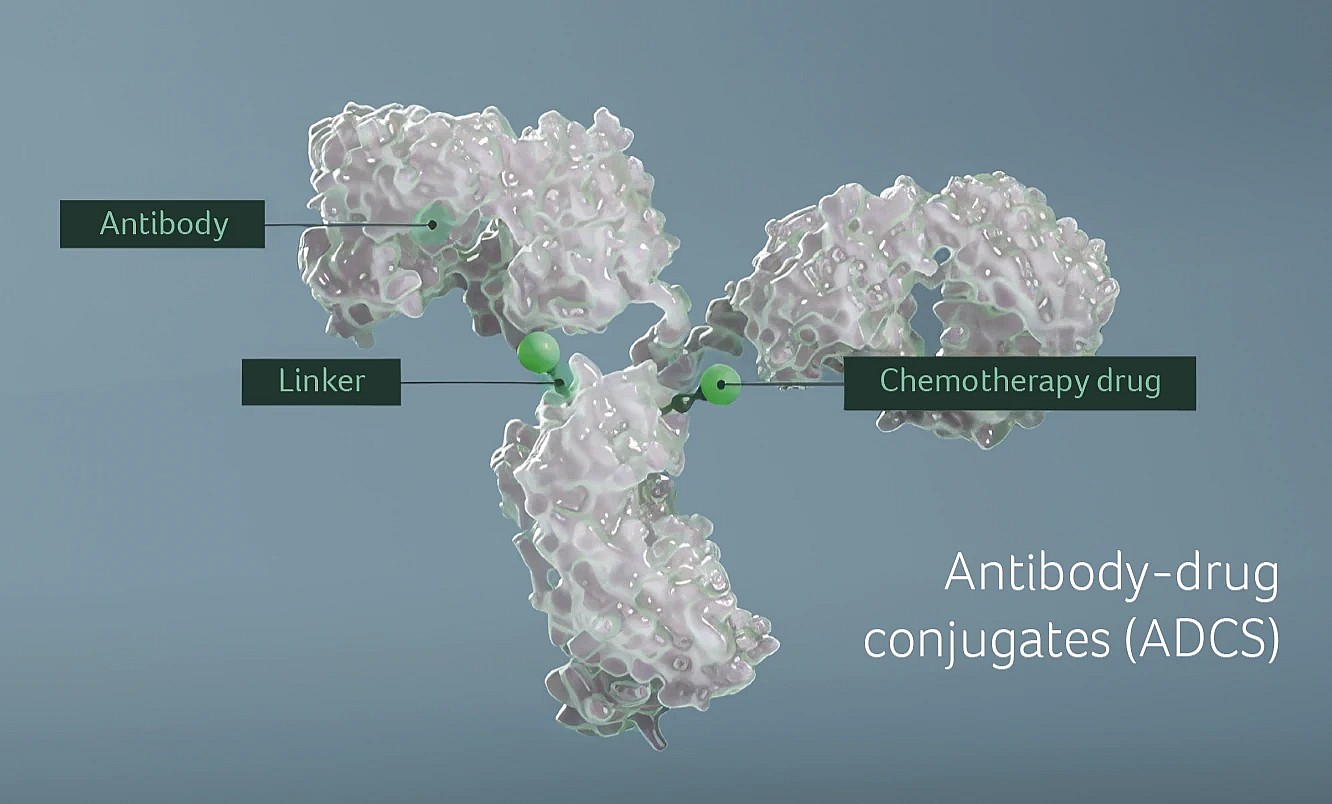
Enhancing Swiss manufacturing capacity and capabilities
Switzerland offers regional, high-tech production of biologics and advanced therapies for global markets, supported by a specialized talent base and a strong regulatory environment. Manufacturing activities range from large-scale commercial supply to highly complex and flexible production of biologics, peptides, and advanced modalities.
Alongside Swiss-based manufacturers such as Lonza and Bachem, which provide mammalian and microbial bioproduction, cell and gene therapy manufacturing, peptide synthesis, and advanced process development, Switzerland hosts significant production activities of multinational pharmaceutical companies including Novartis and Roche. Several international players like Biogen, CSL Behring, Takeda, Johnson & Johnson, and Merck have established manufacturing operations in Switzerland, reflecting the country's role as a trusted location for complex and regulated production.
A key enabler is Switzerland's dual education and vocational training system, which provides a steady pipeline of skilled technicians, laboratory specialists, and engineers trained in GMP environments. These profiles are complemented by process scientists, automation engineers, and data specialists educated at universities and universities of applied sciences, reflecting the increasing technological complexity of modern biomanufacturing.
This combination of vocational and academic talent allows Swiss manufacturing hubs to operate highly regulated, technology-intensive production sites, implement new platforms and modalities, and scale processes efficiently, positioning Switzerland as a location for advanced biologics manufacturing serving global companies.
Commercial HQ — Interview with Alexey Kutahov, Head of International Region, Mirum Pharmaceuticals
For commercial headquarters functions, Switzerland provides a talent environment that supports global leadership, market access, and international coordination. Many biotech companies have chosen Switzerland as a base for commercial and regional headquarters, benefiting from a multilingual workforce, proximity to European markets, and a stable regulatory and political framework.

Could you briefly outline Mirum's core focus areas?
At Mirum, we focus on bringing high impact medicines to patients with rare diseases that have historically been overlooked. We're a global rare disease company, but what truly defines us is how closely our teams are connected to science, patients, and to one another.
The cornerstone of our portfolio of commercial medicines is LIVMARLI®, which is approved for the treatment of cholestatic pruritus in patients with Alagille syndrome (ALGS) and progressive familial intrahepatic cholestasis (PFIC). In these conditions, impaired bile flow leads to bile acid buildup and debilitating symptoms, severely impacting patients' lives.
Alongside our approved medicines, we continue to invest in the next generation of Mirum medicines, with a pipeline of late-stage programs across rare liver and neurological diseases which we believe have the potential to meaningfully improve patients' health and wellbeing.
What were the key reasons for establishing Mirum's commercial headquarters in Switzerland, and what role did the local talent ecosystem play in that decision?
Switzerland was a deliberate choice for Mirum's global commercial headquarters because it sits at the center of one of the world's strongest life sciences ecosystems. This country brings together global pharmaceutical leaders, innovative mid-sized companies, and emerging biotechs, creating a dynamic environment and a local talent pool with experience across the full lifecycle of medicines. That breadth of experience is especially important for a rare disease company like Mirum, where launches and long-term patient support require specialized knowledge, and close collaboration. Switzerland is also ideally positioned geographically, enabling close engagement with clinical sites, physicians, and patient communities across Europe.
What is Mirum's broader European and international growth strategy?
Mirum's European and international growth strategy is focused on expanding access to our medicines while building a sustainable global footprint.
Our commercial approach is intentionally multifaceted. We commercialize directly in North America and Western Europe, where we have established capabilities, and partner strategically in other regions to efficiently reach patients globally.
At the same time, we are focused on advancing a robust late-stage pipeline with global potential. A key near term catalyst will be for brelovitug, which we recently added through our acquisition of Bluejay Therapeutics and are developing for chronic hepatitis delta virus, the most severe form of viral hepatitis. Brelovitug has received EMA PRIME designation, and we expect topline Phase III data from our EU registrational AZURE-2 and AZURE-3 studies in the first half of 2028.
Today, what specific advantages does Switzerland's life sciences ecosystem offer when it comes to attracting, developing, and retaining senior commercial and executive talent?
I have been part of the life sciences community in Switzerland for more than 12 years and have also worked across other European life science hubs during my 25-year career in pharmaceuticals.
"What stands out to me about Switzerland is the depth and maturity of its commercial and executive talent. Leaders here gain exposure to complex launches, global market access, and multiple therapeutic areas, while also having the opportunity to grow their scope and impact without needing to move between regions — which is critical for long‑term retention."
Combined with strong international connectivity and a high quality of life that appeals to globally mobile leaders and their families, Switzerland enables companies like Mirum to attract experienced talent, continue developing leadership capabilities, and retain senior teams over time.
As Mirum continues to scale internationally, how do you see the role of Switzerland evolving?
As our company continues to scale, Switzerland will remain our European and international base. As our global footprint of approved products expands and our pipeline of investigational medicines advances, we expect our Swiss operations in Zug and Basel to continue growing across essential functions, helping us bring more breakthrough therapies to rare disease patients around the world.