- Get directions
- Leave a review
- Claim listing
- Bookmark
- Share
- Report
- prev
- next
- Tuesday, May 5, 2026 @ 11:00 am
While 2025 remained a challenging year for the global biotech sector due to ongoing challenges in the financing environment of public capital markets, Switzerland continued to buck the trend, with new record revenues (CHF 7.5 billion) and R&D investments remaining at very high levels (CHF 2.5 billion in 2025 compared to CHF 2.6 billion in 2024). In terms of financing, Swiss biotech companies raised CHF 2.6 billion (an increase of 2.1% over 2024), but with one important shift in gravity: privately funded biotech companies raised a new record of CHF 1.15 billion, up 38% compared to 2024, and significantly increased their relative share, now making up 45% of the total funding pot, a testimony to the strong private investor community for Swiss biotech.


Similar to 2023 and 2024, 2025 continued to be a difficult year for the biotech sector overall, mainly due to ongoing challenges in the financing environment of public capital markets. Globally there were only 16 biotech IPOs in 2025 (2024: 30), generating approximately USD 2.5 billion in funds (2024: USD 4.0 billion). Of the IPO class of 2025, 12 were in the US, two in Israel, one in the UK, and one in Switzerland, which was BioVersys' successful IPO on SIX Swiss Exchange in February 2025 raising CHF 80 million, marking the largest biotech IPO in Europe in five years. Please refer to additional details about the BioVersys IPO as part of the SIX interview.
Despite the difficult capital market environment, many Swiss biotech companies, especially the privately funded companies, developed strongly in 2025, with the most noteworthy headline that privately funded companies achieved a new record in financing, raising in total CHF 1.15 billion, an increase of 38% compared to 2024. Private company financings in 2025 made up 45% of the total funds raised by Swiss biotech companies, a significant jump compared to the financing share of privately-funded companies in prior years, which was usually around 30% or lower.
Swiss biotech landscape
In 2025, the Swiss biotech industry overall saw a new record in revenues (CHF 7.5 billion in 2025 compared to CHF 7.2 billion in 2024), whereas R&D investments slightly decreased (CHF 2.5 billion in 2025 compared to CHF 2.6 billion in 2024). Also, as a sign of the vibrant Swiss biotech sector, the overall number of FTEs working in Swiss R&D biotech companies increased to a new record of more than 21,000 employees. The increase in FTEs should be seen in the context of more and more companies having advanced to commercial territories, as well as the increased relevance of Swiss CDMOs.
As CDMO activity today includes increasingly complex molecular structures, combined with more rigorous GMP regulations, the specialized R&D and production know-how of Swiss CDMOs have seen a significant uptick in demand. Also, looking at the performance of public and private biotech companies separately, it is impressive how well private biotech companies performed in 2025, with record levels of financing of CHF 1.15 billion (2024: CHF 833 million). Privately financed biotech companies, although R&D expenses remained stable at CHF 1.4 billion in 2025, also set a new record in terms of revenues, with in total CHF 2.1 billion, an increase of 4% compared to 2024, again evidencing how more companies have advanced into the commercial space and are delivering strong growth.
In terms of overall financing, the Swiss biotech industry raised 2.6 billion in 2025 (2024: CHF 2.5 billion), a 2.1% increase compared to 2024, with around CHF 1.4 billion collected by public companies and CHF 1.15 billion collected by private companies. Windward Bio (with CHF 186 million raised) and GlycoEra (with CHF 104 million raised) were the two largest private company financing transactions in 2025, contributing to the new record level of financing for private Swiss biotech companies.
As mentioned above, given the overall difficult capital market environment, there was only one Swiss listing activity in 2025 (BioVersys) which was the only biotech IPO in Europe in 2025. Further, MoonLake Immunotherapeutics in 2025 secured USD 575 million (CHF 490 million) in two separate financing transactions — first a non-dilutive financing facility from Hercules Capital of USD 500 million in April 2025, and later a follow-on financing for an additional USD 75 million, to fund operating expenses and capital expenditure requirements into the second half of 2027.
In summary, while 2025 was a difficult year in terms of capital markets, and some companies also had to initiate restructuring measures, many companies were able to navigate the difficult environment well and were still able to hit significant development or product approval milestones in 2025. Particularly interesting in 2025 was that these important approval milestones are no longer solely European or US approvals, but increasingly also cover other significant markets (e.g., China). Also, while for private biotech companies total capital investments in 2025 reached a new record, it should be noted that, as in 2024, only relatively few companies benefited, with many companies having received little or no funding in 2025. This is also evidenced by the fact that the total financing of the Top 5 financing transactions in 2025 increased by 9% for private biotech companies, compared to 2024.
In addition to, or one could say as a result of, capital markets being more difficult to access for many companies, Swiss biotech companies continued to be agile in terms of finding alternative ways of financing (licensing, R&D collaborations, but also monetization of assets transactions) which, in a similar way to 2024, provided significant non-dilutive financing.
Major 2025 public financing transactions
| Public companies | CHF million |
|---|---|
| Moonlake Immunotherapeutics | 490 |
| Oculis | 274 |
| Idorsia | 216 |
| ADC Therapeutics | 129 |
| Bioversys | 80 |
| Total | 1,189 |
Major 2025 private financing transactions
| Private companies | CHF million |
|---|---|
| Windward Bio | 186 |
| GlycoEra | 104 |
| Orbis Medicines | 84 |
| Nuclidium | 79 |
| Rhygaze | 78 |
| Total | 531 |
M&A and collaborations
Swiss companies were involved in several significant M&A transactions in 2025:
In September 2025, INTEGRA Biosciences Group acquired a minority stake in CleanNA to establish a strategic partnership, aimed at strengthening the collaboration in molecular biology and expanding CleanNA's global reach. Further, in December 2025 Relief Therapeutics and NeuroX completed a reverse merger to form MindMaze Therapeutics, a SIX publicly listed company, focused on AI-driven rehabilitation and neurology combining pharmaceutical treatments with digital neurotherapeutics to treat neurological disorders.
At the same time, similar to 2024, entering into new collaboration and licensing agreements was important for Swiss biotech companies, and even increased in terms of the deals entered into compared to 2024, with some of those partnerships containing significant financial components, which provided alternative ways of funding. A selection of such collaboration and licensing transactions is shown below:
- AB2 Bio signed an option and licensing agreement with Nippon Shinyaku for Nippon Shinyaku to receive an option to acquire exclusive US rights to commercialize Tadekinig alfa to treat primary monogenic IL-18 driven hyperinflammatory syndrome, a rare and potentially life-threatening pediatric disease.
- BioVersys and Shionogi entered into a global research and exclusive license option agreement to jointly develop novel ansamycin leads from BioVersys' BV500 program into clinical candidates, with BioVersys to receive upfront and near-term research payments of CHF 5 million and, upon exercise of the license option, regulatory and sales milestones of up to CHF 479 million as well as royalties on future sales.
- Basilea in-licensed ceftibuten-ledaborbactam etzadroxil, a Phase III-ready oral BL/BLI antibiotic for multidrug-resistant cUTI, from Venatorx, strengthening its late-stage pipeline.
- Debiopharm licensed SunRock's HER3/HER2 bispecific antibody to develop Debio 2512, a next-gen ADC using its MultiLINK™ technology to tackle resistant HER2-driven cancers.
- Boehringer Ingelheim and CDR-Life expanded their collaboration efforts with a global licensing agreement for CDR111, an antibody-based trispecific M-gager® for autoimmune diseases, providing funds to CDR-Life of up to USD 456 million.
- Windward Bio in December 2025 signed a licensing agreement with Qyuns Therapeutics for the development and commercialization of WIN027, a highly potent, long-acting bispecific antibody with best-in-disease potential in respiratory and dermatology diseases. With a deal of up to USD 700 million, this licensing agreement will allow Windward Bio to develop and commercialize WIN027 globally, excluding China.
Product developments
In 2025, the industry saw fewer regulatory approvals, following the record approvals in 2024. The EMA approved 104 new drugs in 2025 (2024: 114 new drugs); similarly, there was a slight decrease in FDA approvals (46 new drugs approved compared to 50 in 2024).
- Idorsia's QUVIVIQ expanded into China as Simcere received NDA approval — Idorsia and Simcere updated their licensing agreement.
- Biocartis received FDA approval for the Idylla™ CDx MSI Test.
- Santhera announced approval in Canada for AGAMREE® (vamorolone) as a treatment for Duchenne muscular dystrophy.
- Altamira Therapeutics announced market approval of Bentrio nasal spray in China.
It is particularly interesting that key approval milestones for Swiss biotech companies do not just center around European and US approvals anymore. Instead, in 2025 more key approvals were also obtained in other significant markets, such as China and Canada. In terms of new product approvals in Switzerland (approvals by Swissmedic), a similar trend could be observed. In 2025, Swissmedic approved 40 new products (2024: 46 approved products).
Awards
Several Swiss biotech companies also received various prestigious awards throughout 2025. These awards included:
- CUTISS was awarded 2nd place at the 2025 edition of The Spark Award – Der Deutsche Digitalpreis, which recognizes outstanding deep-tech innovation across Germany, Austria, and Switzerland.
- Idorsia's treatment for insomnia disorder won the inaugural "Prix Galien Bridges Award" in the "Best Biotechnology & Pharmaceutical Product" category.
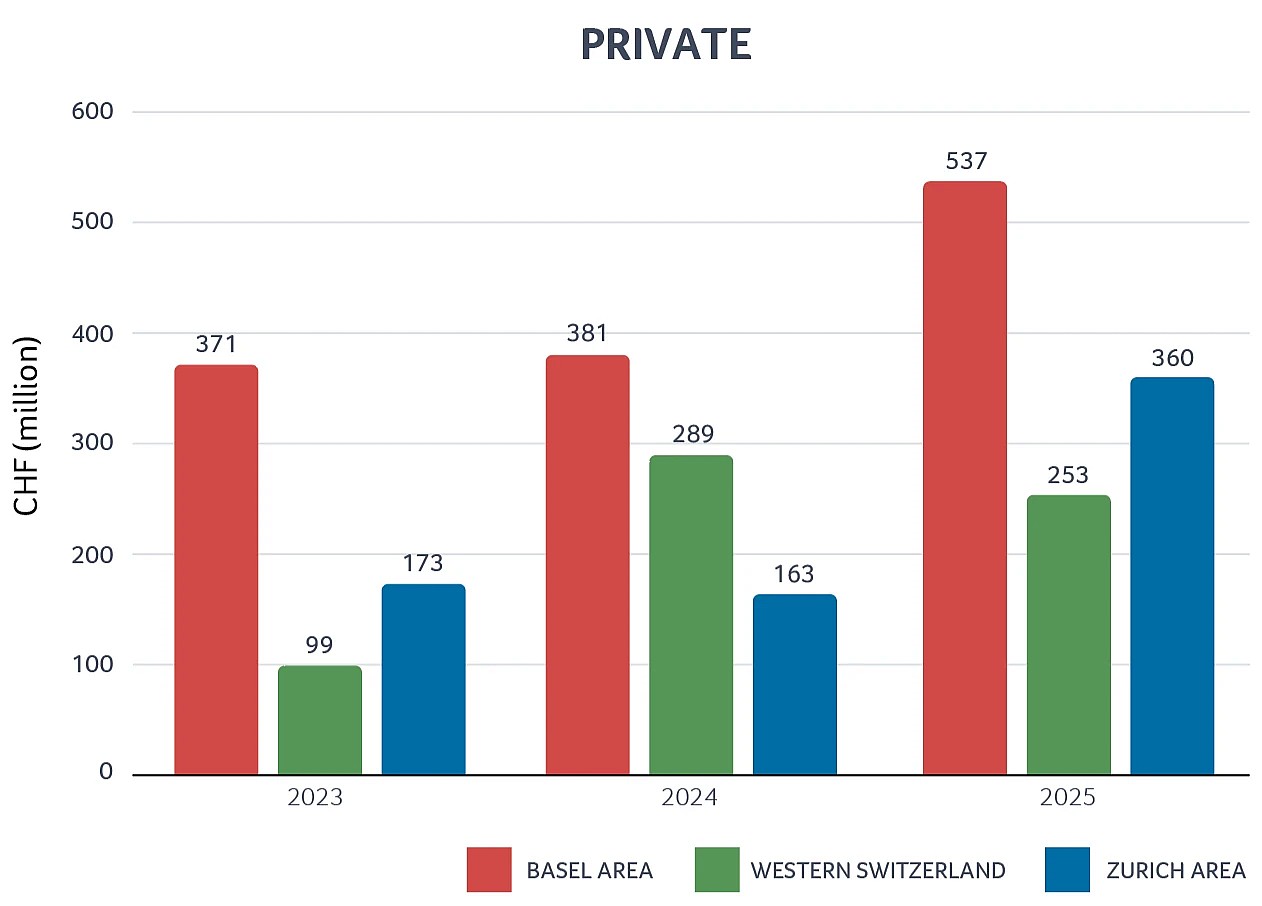
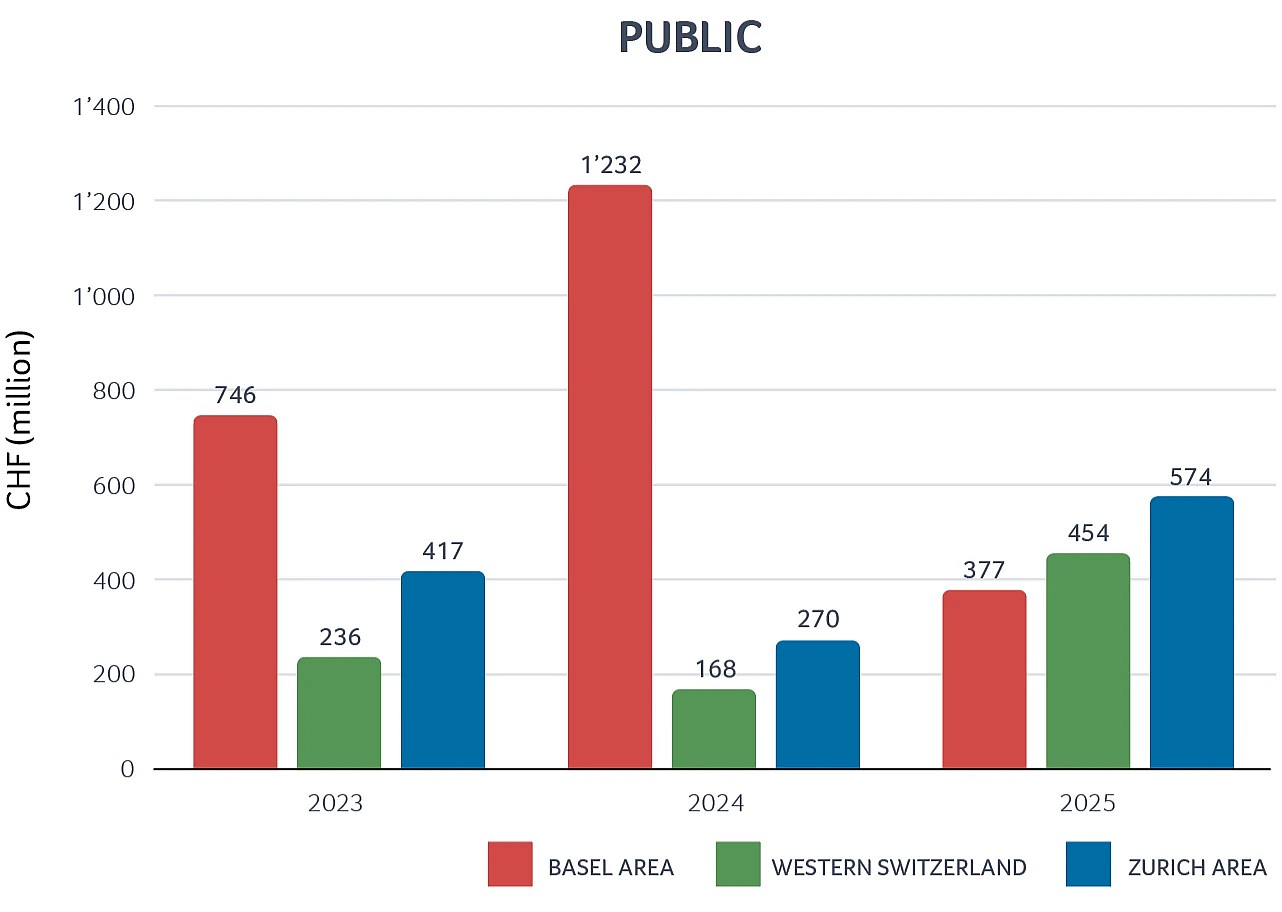
Private & public Swiss biotech regional financing 2023–2025